First article: Effect of Hydrothermal Conditions on Structure of TNT
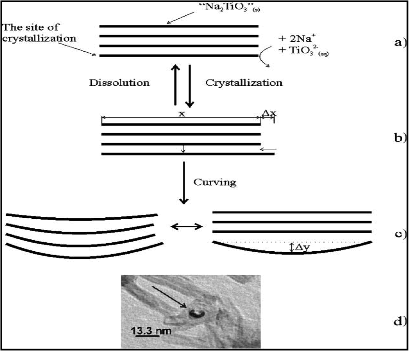
The first article is entitled, “The effect of hydrothermal conditions on the structure of TiO2 nanotubes” by Dmitry V. Bavykin, Valentin N. Parmon, Alexei A. Lapkin and Frank C. Walsh, a combined team of chemical engineers from the University of Bath, Novosibirsk State University and University of Southampton. This paper, which was published in the Journal of Materials Chemistry by The Royal Society of Chemistry, analyzes the possible methods in synthesizing TiO2 nanotubes and aim to explain the mechanism behind the alkali hydrothermal method. Through its findings, the article also discusses about how each preparation condition can affect the morphology of TiO2 nanotubes formed (e.g. diameter and length, wall thickness, pore size). One of their key finding is that “the driving force for wrapping is considered to be the mechanical stress arising during crystallisation/dissolution”. To understand this, they presented a schematic diagram as shown in Figure 1 below.
The first article is entitled, “The effect of hydrothermal conditions on the structure of TiO2 nanotubes” by Dmitry V. Bavykin, Valentin N. Parmon, Alexei A. Lapkin and Frank C. Walsh, a combined team of chemical engineers from the University of Bath, Novosibirsk State University and University of Southampton. This paper, which was published in the Journal of Materials Chemistry by The Royal Society of Chemistry, analyzes the possible methods in synthesizing TiO2 nanotubes and aim to explain the mechanism behind the alkali hydrothermal method. Through its findings, the article also discusses about how each preparation condition can affect the morphology of TiO2 nanotubes formed (e.g. diameter and length, wall thickness, pore size). One of their key finding is that “the driving force for wrapping is considered to be the mechanical stress arising during crystallisation/dissolution”. To understand this, they presented a schematic diagram as shown in Figure 1 below.
|
They proposed that the curving of the TiO2 precursor can be induced by dissolving it in concentrated NaOH solution. After which, a series of reactions occur which lengthens parts of the precursor by crystallizing dissolved TiO2 into nanosheets. As the lengthening process is unequal for the nanosheets, mechanical stress is induced which causes them to conform into a tube like morphology, forming the nanotube. |
This article is highly detailed and well substantiated as seen from the utilization of many formulas such as the Young’s modulus, Thermodynamics equations, equilibria equations. Furthermore, the article managed to accurately explain most of their findings. The article would be better if they had discussed certain possible limitations of this method of synthesis (e.g. loss of uniformity in nanotubes, lower structural stability in terms of tensile strength and durability).
This article had definitely helped us by providing us with details on how to synthesis the TiO2 nanotubes for our project. The article also included certain technical methodologies that have to be in place for a successful synthesis of the nanotubes, thus ensuring that the results obtained would be accurate. For example, it stated that “a temperature from 120⁰C to 150⁰C would allow for nanotube formation” and that “subsequent increases in the temperature result in the formation of non-hollow TiO2 nanofibers”. This information was highly crucial in our methodology as we had to ensure the nanotube morphology of TiO2. Furthermore, the SEM images in the article also provided us a reference to compare with the SEM images of our synthesized nanotubes.
This article had definitely helped us by providing us with details on how to synthesis the TiO2 nanotubes for our project. The article also included certain technical methodologies that have to be in place for a successful synthesis of the nanotubes, thus ensuring that the results obtained would be accurate. For example, it stated that “a temperature from 120⁰C to 150⁰C would allow for nanotube formation” and that “subsequent increases in the temperature result in the formation of non-hollow TiO2 nanofibers”. This information was highly crucial in our methodology as we had to ensure the nanotube morphology of TiO2. Furthermore, the SEM images in the article also provided us a reference to compare with the SEM images of our synthesized nanotubes.