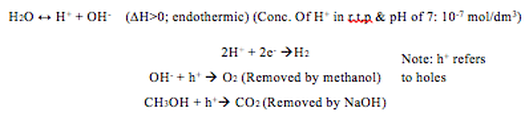Effects of Initial Cu2+ Concentration on Hydrogen Gas Generation
Firstly, to summarise the hydrogen production process, here are the key chemical equations:
The Cu2+ concentration was a major variable for hydrogen generation activity. It can be noted from Figure 8 and Table 3 that the hydrogen evolution increased with the Cu2+ concentration. Within the duration of 60 min, the total amount of hydrogen collected increased drastically, under the effect of Cu2+ concentration, from 6.8ml in the Cu2+-absent sample to 194.7 ml in the 100 mg/L sample.
It is observed that the rate of hydrogen generation is extremely similar across all samples, except for the Cu2+-absent sample, for the first 2 minutes of the experiments, at 3.7 - 4.0ml. Even after 4 minutes, the samples with 10 mg/L and 100 mg/L concentrations were still close in hydrogen generation activity, at 8.0 - 8.2 ml. However, after 8 minutes, there were distinct differences in hydrogen generation activities across all samples. The delay can be attributed to the time needed for the UV light source to stabilize and the initial accumulation of hydrogen gas in the delivery tube. It is also possible that the large presence of Cu2+ in the first 8 minutes acted as a competitive inhibitor for H+ ions, resulting in similar hydrogen gas generation. The Cu2+ concentration gradually dropped as the Cu2+ ions were reduced by electrons, allowing the electrons to reduce more H+.
The 100 mg/L concentration sample, which was the highest initial Cu2+ concentration sample, generated the most hydrogen gas, at 194.7 ml. This was subsequently followed by the 10 mg/L sample and then the 1 mg/L sample, which respectively generated 157.3 ml and 63 ml of hydrogen gas respectively. The Cu2+-absent sample merely generated 6.3 ml of hydrogen gas at the end of the 60 minutes. The results fully support the relationship between initial Cu2+ concentration and the hydrogen generation activity, whereby increasing initial Cu2+ concentrations increases the hydrogen generation activity. Also, this indicates the need for the coexistence of Cu2+ and TiO2 for high photocatalytic activity.
This relationship between the initial Cu2+ concentration and the hydrogen generation activity originate from the fact that the electrons and holes generated during photocatalysis easily recombine together. Hydrogen gas generation during photocatalysis in TiO2 is based upon the photoexcited electron in the valence band of TiO2 being promoted to the conduction band, resulting in a hole in the valence band, which oxidizes OH- ions in water to form oxygen gas, and an electron free to reduce H+ ions in water to form hydrogen gas. However, with the easy recombination of the holes and electrons, it results in low photocatalytic activity, due to the absence of the holes and electrons to oxidize OH- ions and reduce H+ ions in water. The low photocatalytic activity is indicated by the extremely low hydrogen gas generation in the Cu2+-absent sample.
However, with the deposition of Cu2+ on the TiO2 surface, the recombination of the holes and electrons is greatly reduced. With the conduction band of TiO2 higher than that of Cu, the photoexcited electrons in the conduction band of TiO2 can easily transfer to the conduction band of Cu. The addition and accumulation of excess electrons in the conduction band of Cu causes a raise in the Fermi level of Cu. Since a material with a higher Fermi level loses its electrons to another material when they are placed in contact, this allows Cu to be able to act as a water reduction site itself due to rise in Fermi level of Cu from the addition of electrons. This efficient interparticle charge transfer prevents the electron and hole from recombining in TiO2, resulting in high photocatalytic activity as indicated by the higher hydrogen generation activity in the 1 mg/L, 10 mg/L and 100 mg/L samples.
However, it can be observed that the sample with 100 mg/L initial Cu2+ concentration, which adsorbed 9.92 mg of Cu2+, generated far greater hydrogen gas than the sample with 10 mg/L, which adsorbed 9.95 mg of Cu2+. This can be attributed to the possibility of the remaining Cu2+ suspended in the solution also participating in the hydrogen generation and the possibility of UV, being a mechanical stress to TiO2, altering the surface of the TiO2 for greater Cu2+ deposition capacity that would provide more electrons to reduce the H+ ions.
It is observed that the rate of hydrogen generation is extremely similar across all samples, except for the Cu2+-absent sample, for the first 2 minutes of the experiments, at 3.7 - 4.0ml. Even after 4 minutes, the samples with 10 mg/L and 100 mg/L concentrations were still close in hydrogen generation activity, at 8.0 - 8.2 ml. However, after 8 minutes, there were distinct differences in hydrogen generation activities across all samples. The delay can be attributed to the time needed for the UV light source to stabilize and the initial accumulation of hydrogen gas in the delivery tube. It is also possible that the large presence of Cu2+ in the first 8 minutes acted as a competitive inhibitor for H+ ions, resulting in similar hydrogen gas generation. The Cu2+ concentration gradually dropped as the Cu2+ ions were reduced by electrons, allowing the electrons to reduce more H+.
The 100 mg/L concentration sample, which was the highest initial Cu2+ concentration sample, generated the most hydrogen gas, at 194.7 ml. This was subsequently followed by the 10 mg/L sample and then the 1 mg/L sample, which respectively generated 157.3 ml and 63 ml of hydrogen gas respectively. The Cu2+-absent sample merely generated 6.3 ml of hydrogen gas at the end of the 60 minutes. The results fully support the relationship between initial Cu2+ concentration and the hydrogen generation activity, whereby increasing initial Cu2+ concentrations increases the hydrogen generation activity. Also, this indicates the need for the coexistence of Cu2+ and TiO2 for high photocatalytic activity.
This relationship between the initial Cu2+ concentration and the hydrogen generation activity originate from the fact that the electrons and holes generated during photocatalysis easily recombine together. Hydrogen gas generation during photocatalysis in TiO2 is based upon the photoexcited electron in the valence band of TiO2 being promoted to the conduction band, resulting in a hole in the valence band, which oxidizes OH- ions in water to form oxygen gas, and an electron free to reduce H+ ions in water to form hydrogen gas. However, with the easy recombination of the holes and electrons, it results in low photocatalytic activity, due to the absence of the holes and electrons to oxidize OH- ions and reduce H+ ions in water. The low photocatalytic activity is indicated by the extremely low hydrogen gas generation in the Cu2+-absent sample.
However, with the deposition of Cu2+ on the TiO2 surface, the recombination of the holes and electrons is greatly reduced. With the conduction band of TiO2 higher than that of Cu, the photoexcited electrons in the conduction band of TiO2 can easily transfer to the conduction band of Cu. The addition and accumulation of excess electrons in the conduction band of Cu causes a raise in the Fermi level of Cu. Since a material with a higher Fermi level loses its electrons to another material when they are placed in contact, this allows Cu to be able to act as a water reduction site itself due to rise in Fermi level of Cu from the addition of electrons. This efficient interparticle charge transfer prevents the electron and hole from recombining in TiO2, resulting in high photocatalytic activity as indicated by the higher hydrogen generation activity in the 1 mg/L, 10 mg/L and 100 mg/L samples.
However, it can be observed that the sample with 100 mg/L initial Cu2+ concentration, which adsorbed 9.92 mg of Cu2+, generated far greater hydrogen gas than the sample with 10 mg/L, which adsorbed 9.95 mg of Cu2+. This can be attributed to the possibility of the remaining Cu2+ suspended in the solution also participating in the hydrogen generation and the possibility of UV, being a mechanical stress to TiO2, altering the surface of the TiO2 for greater Cu2+ deposition capacity that would provide more electrons to reduce the H+ ions.