Methodology and Justifications
Our project aims to produce hydrogen energy concurrently with heavy metal ions removal from water by TiO2 nanotubes (TNT).
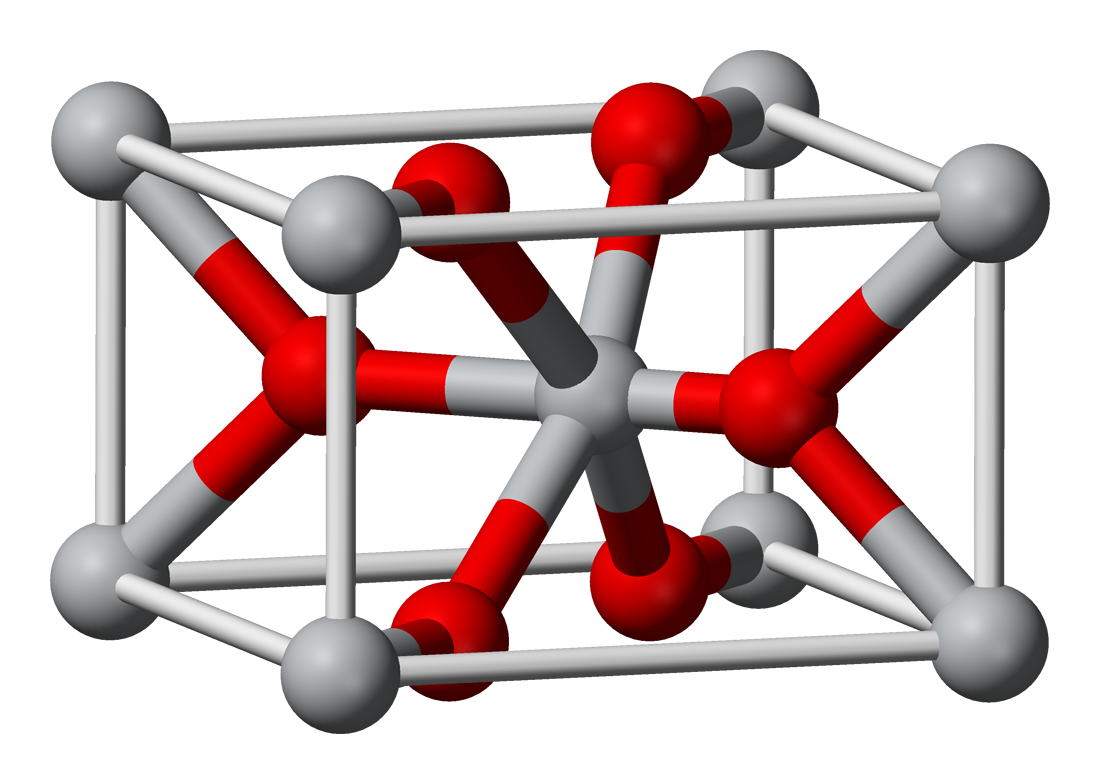
Why TiO2?
We chose TiO2 as it is researched to be “cost-effective, environment-friendly and stable” [2]. Furthermore, TiO2 Ksp is relatively lower than other metal oxides, thus it would be more resilient to acid/basic conditions and be a suitable insoluble material for water-related processes.
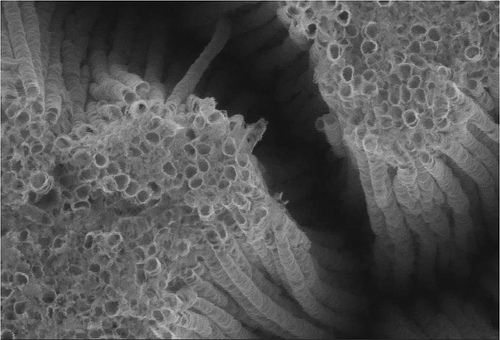
Why nanotubes?
We chose nanotubes over other morphologies as literature has shown that its surface area to volume ratio, 200m2/g, is significantly higher as compared to nanowires or microspheres and we strongly believe that this would have a significant impact on its efficiency. Our fabrication was a suspension rather than a membrane as we contemplated that a membrane would be susceptible to fouling and would restrict the amount of UV irradiation the material can receive.
Why TiO2?
We chose TiO2 as it is researched to be “cost-effective, environment-friendly and stable” [2]. Furthermore, TiO2 Ksp is relatively lower than other metal oxides, thus it would be more resilient to acid/basic conditions and be a suitable insoluble material for water-related processes.
Why nanotubes?
We chose nanotubes over other morphologies as literature has shown that its surface area to volume ratio, 200m2/g, is significantly higher as compared to nanowires or microspheres and we strongly believe that this would have a significant impact on its efficiency. Our fabrication was a suspension rather than a membrane as we contemplated that a membrane would be susceptible to fouling and would restrict the amount of UV irradiation the material can receive.
Why is methanol used in this project?
Our project has also used methanol as the sacrificial reagent during the photocatalysis process as methanol consist of only one carbon and would be theoretically more efficient in resupplying electrons back to the Cu-TiO2 nanotubes efficiently. Furthermore, it was able to react with oxygen present in the reactor, thus creating an anaerobic environment that prevented oxygen gas from reacting with excited electrons which would inhibit hydrogen production. The utilisation of such organic pollutants as sacrificial reagents should not be deemed as wastage of resources as quoted by Prof. Darren Sun, a professional in this field, “small concentrations (around 10 to 20 %) of such organic pollutants in waters are highly difficult to purify” and by utilising it for hydrogen generation, we would be making full use of all resources.
Our project has also used methanol as the sacrificial reagent during the photocatalysis process as methanol consist of only one carbon and would be theoretically more efficient in resupplying electrons back to the Cu-TiO2 nanotubes efficiently. Furthermore, it was able to react with oxygen present in the reactor, thus creating an anaerobic environment that prevented oxygen gas from reacting with excited electrons which would inhibit hydrogen production. The utilisation of such organic pollutants as sacrificial reagents should not be deemed as wastage of resources as quoted by Prof. Darren Sun, a professional in this field, “small concentrations (around 10 to 20 %) of such organic pollutants in waters are highly difficult to purify” and by utilising it for hydrogen generation, we would be making full use of all resources.